Sector(s)
Team Members
About the project
The Challenge
The Clinical Practice Research Datalink (CPRD) manages one of the UK's most valuable healthcare databases, containing anonymised patient records from GP practices across the country. Researchers who want to access this sensitive medical data must submit detailed research proposals that undergo rigorous review by scientific experts and lay reviewers to ensure the research is ethical, scientifically sound, and protects patient privacy.
Previously, this process involved paper forms, email exchanges, and manual coordination between dozens of reviewers, administrators, and researchers. This made it difficult to:
- Track where applications were in the review process
- Ensure consistent review standards
- Maintain clear audit trails
- Communicate efficiently with all parties
- Meet transparency requirements for publishing approved studies
The Solution
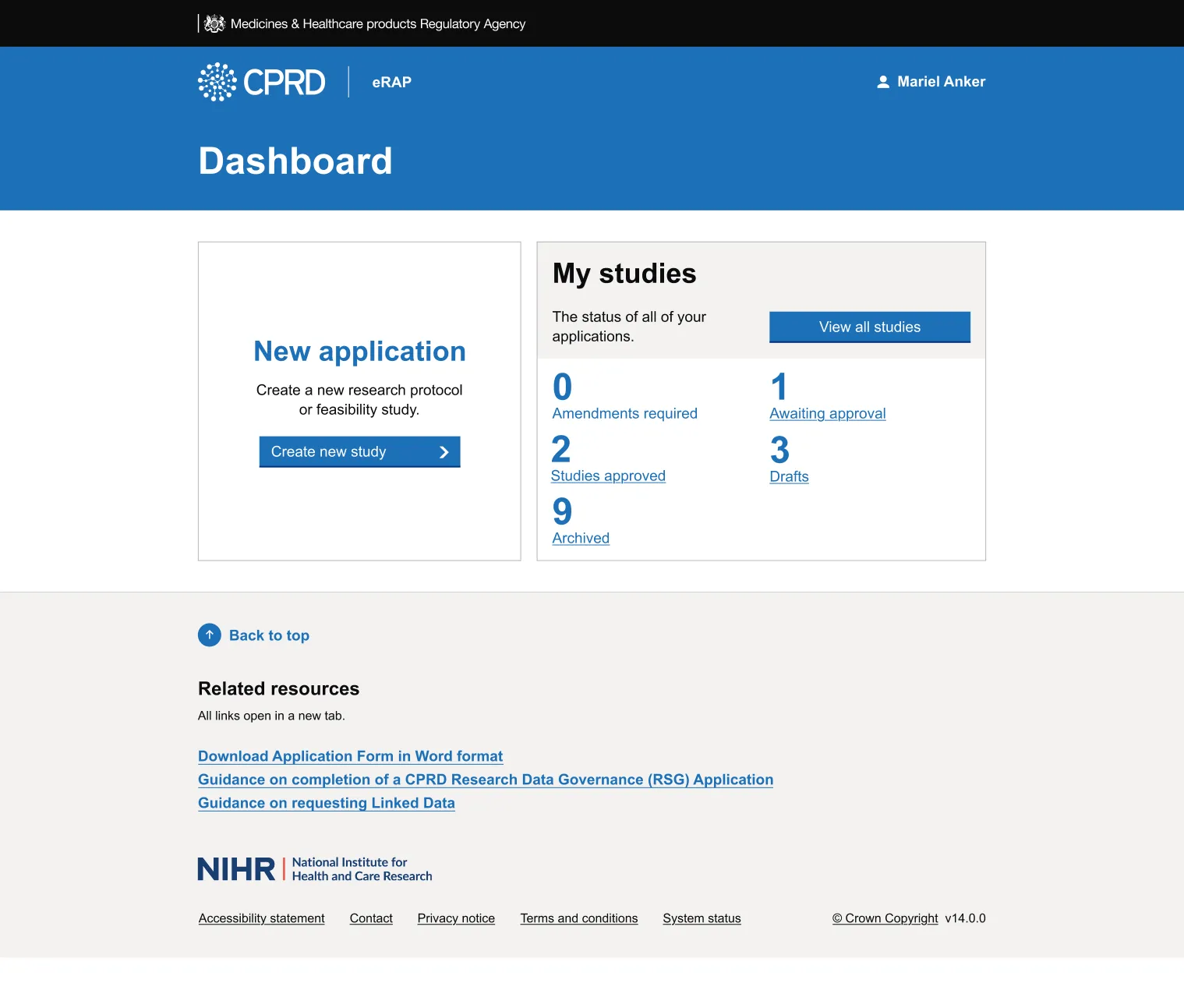
eRAP is a comprehensive online portal that digitises and streamlines the entire research application process from initial submission through final approval. Here researchers can:
- Submit applications through guided online forms that walk them through every required section
- Save drafts and return later to complete their applications
- Track progress through dashboards showing their application status in real-time
- Respond to feedback by submitting amendments when reviewers request changes
- Request data linkages after their main study is approved
For reviewers and administrators, the portal provides:
- Automated workflows that route applications to the right people at the right time
- Structured review forms ensuring consistent evaluation criteria
- Parallel review processes allowing multiple reviewers to work simultaneously
- Clear visibility into all pending tasks and deadlines
- Comprehensive audit trails of all decisions and communications
Key Benefits
For Researchers:
- Simplified submission process with clear guidance
- Transparent status updates at every stage
- Faster turnaround times
- Ability to manage all applications in one place
For CPRD:
- Reduced administrative burden
- Consistent, high-quality reviews
- Full compliance with governance requirements
- Better resource management and workload visibility
- Automatic publication of approved studies for transparency
For the Public:
- Enhanced oversight of how patient data is used
- Lay reviewer involvement in approval decisions
- Public transparency about approved research projects
How It Was Built
Drupal is well-suited for complex, form-heavy applications that require sophisticated workflows and strict access controls. It's pre-built toolkit handles common website needs (user accounts, security, databases) so developers can focus on building the unique features CPRD requires.
The Building Blocks
1. Smart Forms That Guide Users
The application uses intelligent online forms that adapt as users fill them in. The forms:
- Show only relevant questions based on previous answers
- Save progress automatically so nothing is lost
- Validate information immediately to catch errors early
- Provide helpful explanations and examples alongside each question
- Work on any device (computer, tablet, or phone)
These forms are built using Drupal's Webform module, which was extended to meet CPRD's specific needs.
2. Configurable Review Workflows
The CPRD review process involves many parties that review, approve, deny or request amendments to the application. The system:
- Automatically routes applications to the right reviewers
- Tracks who needs to complete which tasks
- Sends reminders when deadlines approach
- Allows for multiple rounds of feedback and revision
- Can handle different review paths for different application types
This flexibility means CPRD can adapt the process as requirements change without needing to rebuild the entire system.
3. Seamless Integration with Existing Systems
Rather than replacing everything CPRD uses, the portal connects to their existing tools:
- Salesforce CRM - Where CPRD already manages customer relationships and generates official documents
- Single Sign-On (via CPRD Gateway) - Allowing users to log in using their institutional credentials rather than creating yet another password
- Notifications systems (via CPRD Gateway) - For sending notifications at appropriate times
These integrations happen behind the scenes, creating a seamless experience where data flows automatically between systems without manual data entry or risk of errors.
4. Role-Based Access Control
Different users see different things based on their role:
- Researchers see their own applications and can submit new ones
- Reviewers see only applications assigned to them with relevant review sections
- Administrators have overview of all applications and can manage assignments
- Moderators coordinate the review process and make final decisions
5. Government Design Standards
The portal follows GOV.UK design patterns, which means:
- Clean, accessible interface that works for users with disabilities
- Plain language instead of jargon
- Mobile-friendly design
- Consistent with other UK government services users may be familiar with
Technical Approach
Modular Design
Rather than building one massive system, the solution is constructed from interconnected modules, each handling a specific function (applications, reviews, notifications, user management, etc.).
Configuration Over Customization
Wherever possible the CPRD staff can adjust workflows, change form questions, or modify review criteria by editing settings rather than requiring developers to write new code.
Automated Testing
The system includes automated testing capabilities, allowing administrators to simulate the entire review process to ensure everything works correctly before real applications go through.
The Result
The Drupal system successfully transformed a complex, paper-based process into a streamlined digital experience. It demonstrates how modern web technology can be applied to highly regulated, sensitive domains like healthcare research governance while maintaining the human oversight and expert judgment that makes the ISAC review process valuable.
The Drupal setup handles the routine tasks (routing, notifications, tracking) automatically while empowering human experts to focus on what they do best: evaluating the scientific merit and ethical implications of proposed research.
Why Drupal was chosen
Drupal was chosen for its ability to support complex, form-driven workflows with robust security, granular permissions and seamless integration with existing government systems.
Technical Specifications
Drupal version:
Key modules/theme/distribution used:
- Complicated workflows captured in Workflow with various external parties providing review and sign off based on the submission.